A structural formula for methane contains symbols for one C atom and four H atoms, indicating the number of atoms in the molecule ( Figure 1). 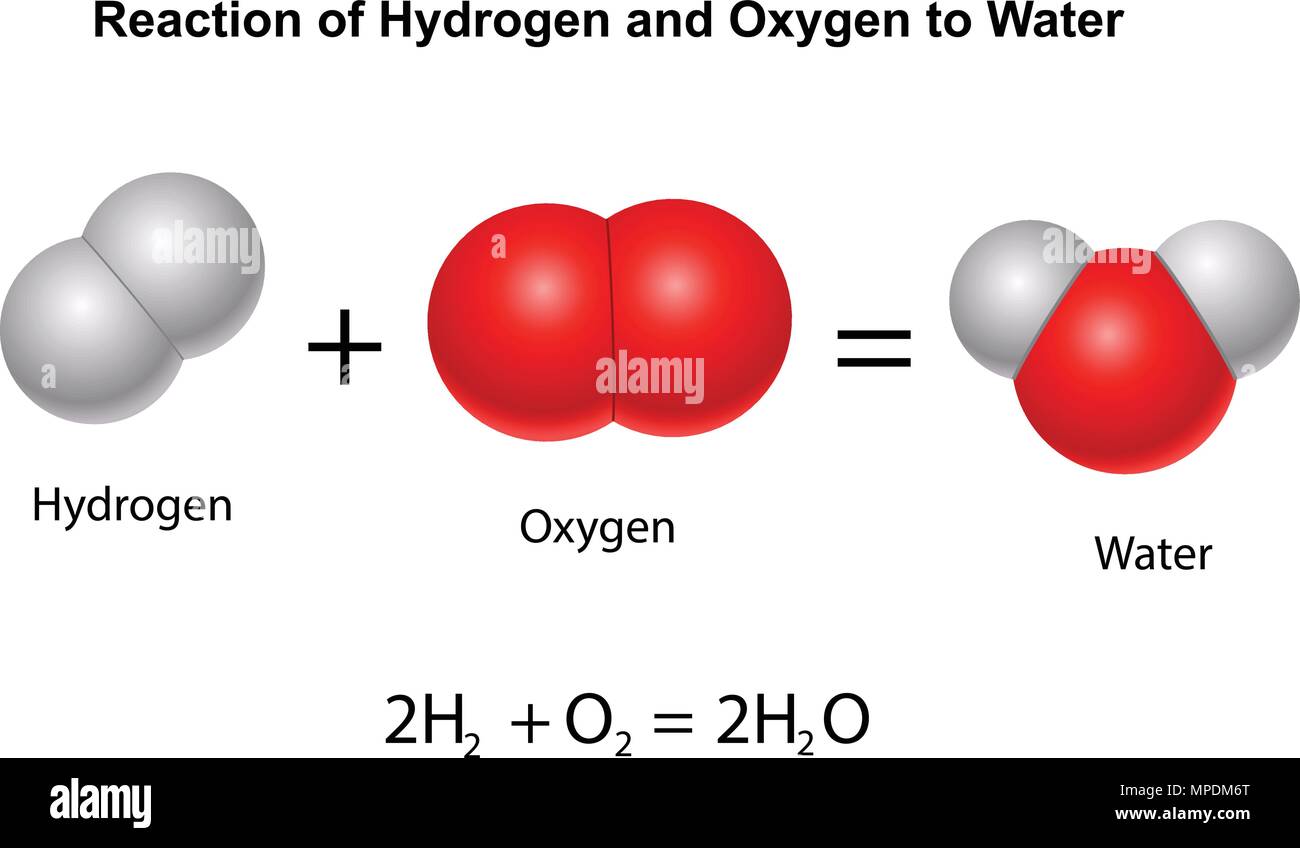 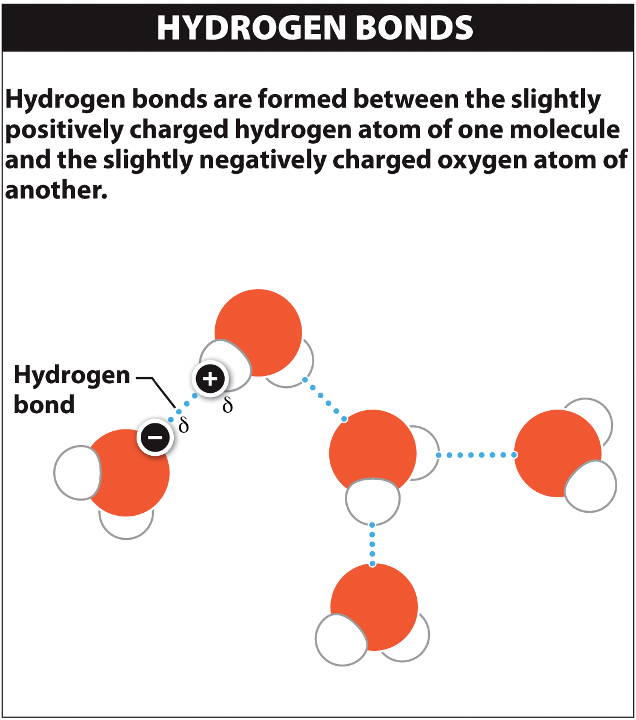
Various types of structural formula for a compound give the same information as the molecular formula, but also shows how the atoms are connected in the molecule. (A subscript is used only when more than one atom of a given type is present.) Molecular formulas are also used as abbreviations for the names of compounds. Now we will investigate some of the ways molecules and compounds are described.Ī molecular formula is a representation of a molecule that uses chemical symbols to indicate the types of atoms followed by subscripts to show the number of atoms of each type in the molecule. We have already been introduced to symbols for elements, atoms, ions and isotopes. Like models we use in other parts of our lives, these representations are helpful to us even if they aren’t exactly like the real thing. Each type of representation has its own level of detail, its own accuracies and inaccuracies. All of the various representations used are models: approximations of the real atoms and molecules under discussion. Because atoms and molecules are too small to see, this language is used extensively to share and discuss chemical ideas. There is a rich and complex symbolic language used in chemistry. Represent the bonding arrangement of atoms within molecules using structural formulas.Symbolize the composition of molecules using molecular formulas.By the end of this section, you will be able to: 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
